Get the ShieldTM for Patient Safety






IMPROVE PATIENT SAFETY
Shield patients from intra-operative insulation failure and capacitive energy burns, per AORN electrosurgical safety guidelines.
REDUCE OR EXPENSES
Improve business performance with free Shield™ Instruments and reposable 2x Scissors.
BOOST CLINICAL PERFORMANCE
Improve the surgical experience with high performance instrumentation and the sharpest scissors.
"*" indicates required fields
FDA Issues a Safety Communication on the Dangers of Monopolar Laparoscopic Surgery. “Monopolar Energy Use Can Directly Result in Unintended Patient Burns from Capacitive Coupling and Intra-Operative Insulation Failure.”
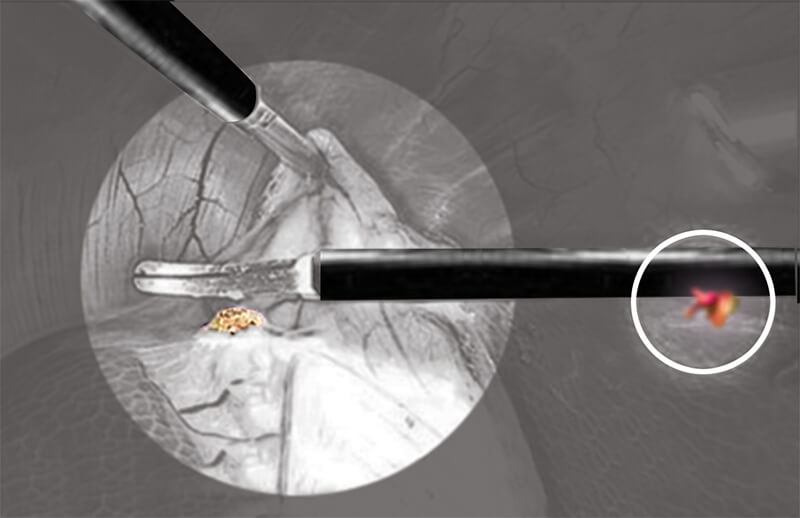
The new AORN Electrosurgical Safety Guideline recommends to “use an Active Electrode Monitoring (AEM®) and Shielding Device” during Minimally Invasive Surgery.

AEM enTouch® Reposable 2x Scissors

Best-in-class performance at an exceptional value
Super sharp and exceptionally responsive, AEM enTouch® Reposable 2x Scissors provide new levels of performance while reducing cost and waste.
- Micro-serrated blades immobilize tissue during dissection for unmatched surgical precision.
- Dramatic Color Indicators show when it’s time for a new scissors (after 2 uses).
- High Performance AEM enTouch® Handle with tactile feedback.
- Shielded AEM® Instruments minimize surgical complications, by patient burns from intraoperative insulation failure and capacitive coupling.1-3.

"*" indicates required fields
AEM® Shield™ Laparoscopic Instruments win Second-Generation Vizient Innovative Technology Contract (MS1306).
- Vizient is the nation’s leading healthcare improvement company, with 97% of academic medical centers and 60% of all acute care hospitals in the USA.
- The contract is based on a recommendation of AEM® Shield™ Laparoscopic Instruments by hospital experts in this category who serve on one of Vizient’s member-led councils.
- There is no other technology on the market that provides the unique patient safety, performance, and value of Shield Laparoscopic Instruments.

NBC News Investigates: Popular Surgeries Can Carry Risk of Being Burned
AEM® ShieldTM Laparoscopic Instruments highlighted for unique patient safety, in NBC Los Angeles Investigative Report on Stray Energy Burns.
Deliver Surgical Energy Safely.
Improve General Surgery and GYN Outcomes.
- Every 90 minutes in the USA a patient is burned during laparoscopic surgery.
- These preventable stray energy burns kill 1 – 2 patients per day*.
- The FDA issued a Safety Communication on these risks from both intraoperative insulation failure and capacitive coupling. Learn More
- AEM® Shield™ Laparoscopic Instruments eliminate these complications, saving patients lives every day.
Dr. Craig Hornbarger shares how he protects his patients with AEM® ShieldTM Laparoscopic Instruments

